 C 2 = Final concentration of new solution.V 1 = Volume of stock solution needed to make the new solution.The following is a brief explanation of some ways of calculating dilutions that are common in biological science Using C 1V 1 = C 2V 2 To make a fixed amount of a dilute solution from a stock solution, you can use the formula: C 1V 1 = C 2V 2where: The Concentration Factor for this problem = aliquot volume/final volume = 0.1/(0.1 + 9.9) = 0.01 or 10 -2 concentration Dilutions: Explanations and Examples of Common Methods There are many ways of expressing concentrations and dilution. The dilution factor is equal to the final volume divided by the aliquot volume: 10 mL/0.1 mL = 1:100 dilution (10 2). The final volume is equal the the aliquot volume plus the diluent volume: 0.1 mL + 9.9 mL = 10 mL. 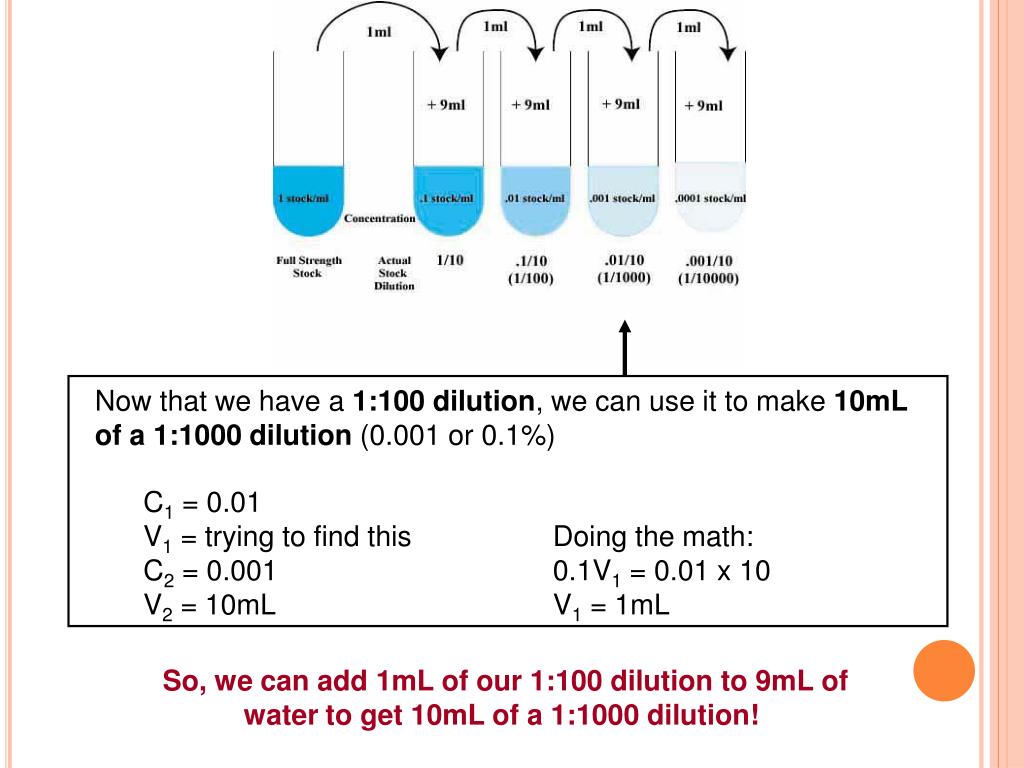
EXAMPLE: What is the dilution factor if you add 0.1 mL aliquot of a specimen to 9.9 mL of diluent? Diluent: material with which the sample is diluted Dilution factor ( DF) : ratio of final volume/aliquot volume (final volume = aliquot + diluent) Concentration factor ( CF) : ratio of aliquot volume divided by the final volume (inverse of the dilution factor) To calculate a dilution factor: Remember that the dilution factor is the final volume/aliquot volume. DEFINITIONS: Aliquot: a measured sub-volume of original sample.  
High dilutions are usually expressed exponentially ( i.e: a solution which has been diluted a million fold is termed a 10 6 dilution, or is 10 -6 concentration). This requires a working knowledge of the principles of diluting, dilution factors, concentration factors and the calculations involved. Because solutions in science are often much more concentrated than are desired or can be managed for a given protocol, it is frequently necessary to dilute these solutions to a desired level. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
